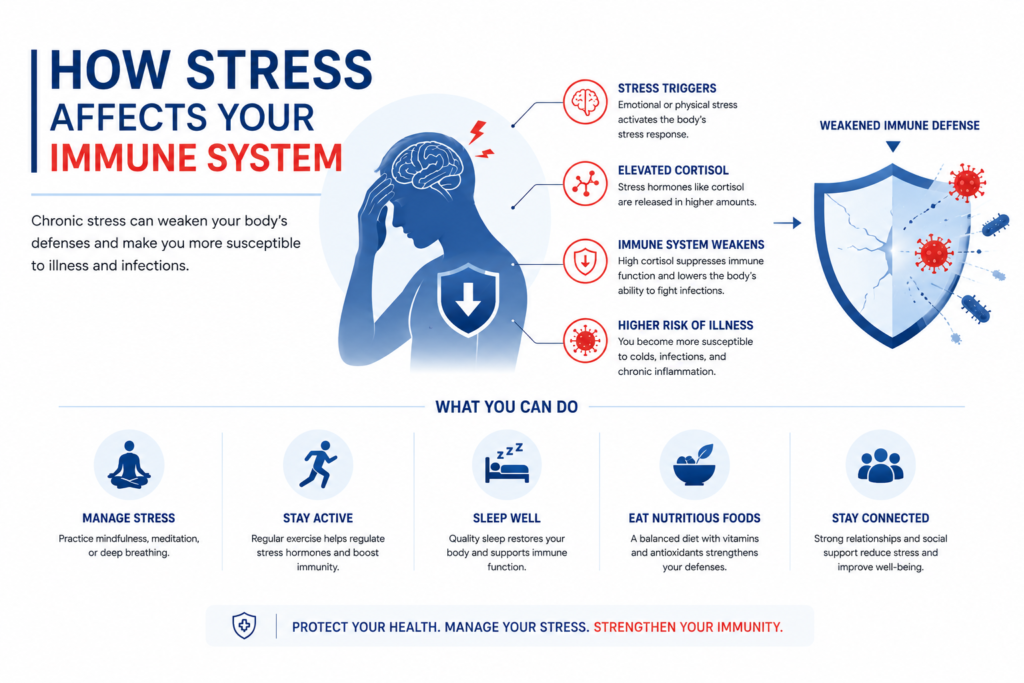
The modern world is characterized by an unprecedented level of constant, low-grade tension. From demanding work schedules and financial pressures to the endless stream of news and the complexities of personal relationships, the human experience is increasingly defined by psychological strain. We often describe ourselves as feeling “burned out,” “overwhelmed,” or simply “stressed.” But what exactly does this mean for our physical health? The commonsense notion that “too much stress makes you sick” has been passed down through generations as conventional wisdom. Today, however, this colloquial phrase is no longer just an observation; it is a scientifically validated reality backed by decades of rigorous medical research.
To understand the profound connection between our emotional state and our physical health, we must venture into the intricate, microscopic world of human biology. Stress is not merely a fleeting emotion or a subjective feeling of unease; it is a powerful, systemic, and highly orchestrated physiological cascade that can fundamentally rewire the body’s defense mechanisms. At the very center of this biological storm is the immune system—the highly complex network of cells, tissues, and organs dedicated to defending the body against pathogens, viruses, bacteria, and even cancerous mutations.
When we experience psychological duress, our brain initiates a chemical and hormonal reaction that reverberates throughout every system in the body. In the short term, this response is a survival mechanism, a brilliant evolutionary adaptation designed to keep us alive in the face of acute danger. However, when this response is chronically activated—as it so often is in modern society—it begins to dismantle the very immune defenses it was designed to support.
This comprehensive guide will take a deep dive into the fascinating world of psychoneuroimmunology, exploring the anatomy of stress, the architecture of the immune system, the cellular mechanisms that connect them, and actionable strategies to protect your health.
The Anatomy of Stress: How the Body Responds
Before we can understand how stress impacts immunity, we must first define what stress is from a biological standpoint. In the realm of physiology, stress is defined as an organism’s total response to an environmental condition or stimulus (a stressor) that disrupts homeostasis, the body’s natural state of internal equilibrium. When a stressor is perceived—whether it is a physical threat like a predatory animal or a psychological threat like an impending deadline—the body responds by stimulating the nervous, endocrine, and immune systems.
The human body relies on two primary physiological superhighways to process and respond to stress: the Sympathetic-Adrenal-Medullary (SAM) axis and the Hypothalamic-Pituitary-Adrenal (HPA) axis.
The Sympathetic-Adrenal-Medullary (SAM) Axis
The SAM axis represents the body’s immediate, short-term response to stress, commonly known as the “fight-or-flight” response. When your brain perceives a threat, a tiny almond-shaped structure called the amygdala sends an alarm signal to the hypothalamus, the brain’s command center. The hypothalamus instantly activates the sympathetic nervous system, sending nerve impulses down the spinal cord and directly into the adrenal glands, which sit atop the kidneys.
Within seconds, the adrenal medulla pumps catecholamines—specifically adrenaline (epinephrine) and noradrenaline (norepinephrine)—into the bloodstream. This surge of adrenaline causes a rapid physiological transformation: your heart rate accelerates to pump more oxygenated blood to your muscles, your airways dilate to take in more oxygen, your pupils dilate to sharpen your vision, and your liver releases stored glucose to provide an immediate burst of energy. Blood is shunted away from non-essential functions like digestion and directed toward the skeletal muscles. This response is lightning-fast, preparing the organism to either fight the threat or run for its life.
The Hypothalamic-Pituitary-Adrenal (HPA) Axis
If the perceived threat does not immediately dissipate, the body engages its second, longer-term stress response system: the Hypothalamic-Pituitary-Adrenal axis. This system operates on hormonal signaling rather than immediate nerve impulses, making it slower to activate but much longer-lasting in its effects.
The HPA axis operates through a complex cascade of chemical messengers:
- The Hypothalamus: Continues the stress response by releasing Corticotropin-Releasing Hormone (CRH).
- The Pituitary Gland: CRH travels a short distance to the pituitary gland, stimulating it to release Adrenocorticotropic Hormone (ACTH) into the systemic blood circulation.
- The Adrenal Cortex: ACTH travels through the blood to the outer layer of the adrenal glands (the adrenal cortex), triggering the synthesis and release of glucocorticoids, the most famous of which is cortisol.
Cortisol is universally known as the primary “stress hormone.” Its job is to keep the body on high alert and provide the sustained energy needed to deal with a prolonged threat. It does this by stimulating gluconeogenesis (the production of new glucose in the liver), increasing fat metabolism, and suppressing physiological processes that are deemed biologically costly and non-essential during a crisis. Crucially, one of the most energy-demanding systems in the human body is the immune system. Consequently, high levels of cortisol profoundly suppress immune function and inflammatory responses to conserve energy for immediate survival.
Table 1: Key Stress Hormones and Their Systemic Effects
| Hormone | Origin | Speed of Release | Primary Physiological Actions |
| Adrenaline (Epinephrine) | Adrenal Medulla | Seconds (Immediate) | Increases heart rate, elevates blood pressure, dilates airways, mobilizes immediate glucose stores. |
| Noradrenaline (Norepinephrine) | Sympathetic Nerves | Seconds (Immediate) | Induces widespread blood vessel constriction, redirects blood flow to essential organs and muscles. |
| Cortisol | Adrenal Cortex | Minutes to Hours | Increases blood glucose via gluconeogenesis, alters metabolism, drastically suppresses immune and inflammatory responses. |
| CRH (Corticotropin-Releasing Hormone) | Hypothalamus | Minutes | Triggers the pituitary gland; also acts within the brain to increase anxiety and decrease appetite. |
The Immune System: An Essential Overview
To fully grasp the damage that unchecked stress hormones can inflict, we must take a tour of the immune system. The immune system is not a single organ but an incredibly diverse, distributed network of specialized cells, tissues, proteins, and organs working in seamless harmony to distinguish “self” from “non-self” and eliminate the latter. The immune system is broadly divided into two main branches: the innate immune system and the adaptive immune system.
Innate Immunity: The First Responders
The innate immune system is your body’s first line of defense. It is primitive, fast-acting, and non-specific, meaning it attacks any foreign invader it encounters without needing to recognize precisely what it is.
Key players in the innate immune system include:
- Physical Barriers: The skin, mucous membranes, stomach acid, and enzymes in tears.
- Phagocytes (Macrophages and Neutrophils): These are the “pac-men” of the immune system. They patrol the body, identify foreign particles or dead cells, engulf them, and digest them in a process called phagocytosis.
- Natural Killer (NK) Cells: A specialized type of white blood cell that roams the body looking for cells that have been infected by viruses or have turned cancerous. When an NK cell finds a compromised cell, it injects it with toxic enzymes, forcing the infected cell to undergo apoptosis (programmed cell death).
- Cytokines: These are the chemical messengers of the immune system. When a macrophage encounters a pathogen, it releases pro-inflammatory cytokines (such as Interleukin-1, Interleukin-6, and Tumor Necrosis Factor-alpha) to sound the alarm, dilate blood vessels, and recruit other immune cells to the site of infection. This creates the localized heat, swelling, and redness we recognize as inflammation.
Adaptive Immunity: The Specialized Assassins
If a pathogen manages to evade the innate immune system, the adaptive immune system is called into action. This system is slower to mobilize (taking days or even weeks to fully activate) but is highly specific and, most importantly, possesses immunological memory.
The adaptive immune system relies primarily on lymphocytes, a type of white blood cell generated in the bone marrow:
- B-Cells (Humoral Immunity): B-cells are responsible for producing antibodies. When a B-cell encounters a specific pathogen (like the influenza virus), it matures into a plasma cell and begins pumping out millions of Y-shaped proteins called antibodies. These antibodies lock onto the specific virus, neutralizing it and tagging it for destruction by macrophages.
- T-Cells (Cell-Mediated Immunity): T-cells mature in the thymus and come in several varieties.
- Helper T-Cells (CD4+): The generals of the immune army. They do not kill pathogens directly; instead, they release specific cytokines to direct and amplify the activity of B-cells, macrophages, and other T-cells.
- Cytotoxic T-Cells (CD8+): The specialized assassins that actively seek out and destroy cells that have been hijacked by viruses.
- Regulatory T-Cells (Tregs): The peacekeepers. Once an infection is cleared, Tregs suppress the immune response to prevent the system from attacking the body’s own healthy tissues (autoimmunity).
The balance between all these cells, orchestrated by the delicate language of cytokines, is vital for human health. As we will see, stress disrupts this delicate communication network entirely.
The Birth and Evolution of Psychoneuroimmunology
For centuries, Western medicine operated under the assumption of Cartesian dualism—the philosophical belief proposed by René Descartes that the mind and the physical body are entirely separate entities. Under this paradigm, it was believed that thoughts, emotions, and psychological stress could not possibly influence physiological systems as distinct as the immune response.
This dogma was shattered in the late 20th century, giving rise to an entirely new, revolutionary field of medicine: Psychoneuroimmunology (PNI).
PNI is the interdisciplinary study of the complex interactions between psychological states, the central nervous system, the endocrine (hormone) system, and the immune system. The foundational premise of PNI is that there is no real division between the mind and the body. Instead, they are connected by vast, bidirectional networks of communication.
The Seminal Discoveries
The field began to take shape in the 1970s and 1980s through a series of groundbreaking experiments. One of the most famous was conducted by psychologist Robert Ader and immunologist Nicholas Cohen in 1975. In a classic Pavlovian conditioning experiment, they gave rats saccharin-sweetened water simultaneously paired with cyclophosphamide, a powerful drug that induces nausea and suppresses the immune system. Eventually, they stopped giving the drug and only gave the rats the sweetened water. Astoundingly, the rats’ immune systems still crashed. The researchers had proven that the immune system could be classically conditioned by the brain. The hierarchical model was clear: neural impulses from a taste stimulus could completely alter immune function.
Bidirectional Communication: The Brain-Body Dialogue
Research in PNI has elegantly mapped out exactly how the brain and the immune system “talk” to one another.
- Brain to Immune System: The brain communicates with the immune system via the SAM and HPA axes. Furthermore, researchers discovered that primary and secondary lymphoid organs (like the spleen, thymus, lymph nodes, and bone marrow) are heavily innervated by the autonomic nervous system. Nerve fibers literally plug directly into immune tissues, allowing the brain to release neurotransmitters straight onto immune cells.
- Immune System to Brain: The communication goes the other way, too. When the immune system detects an infection, it produces pro-inflammatory cytokines. These cytokines can travel through the bloodstream, cross the highly selective Blood-Brain Barrier (BBB), and bind to receptors in the brain. This triggers what scientists call “sickness behavior”—the lethargy, depression, social withdrawal, and loss of appetite you experience when you have the flu. The immune system is essentially telling the brain to force the body to rest and conserve energy to fight the infection.
The Rise of Immunopsychiatry
In recent years, the findings of PNI have birthed a sub-discipline known as immunopsychiatry. This field explores how chronic, low-grade inflammation in the body can actually cause psychiatric conditions like major depressive disorder and anxiety. Studies have shown that individuals with high levels of systemic inflammation are at a significantly higher risk of developing depression, highlighting that emotional distress can be a symptom of an overworked, stressed immune system.
Acute vs. Chronic Stress: The Dual Nature of the Immune Response
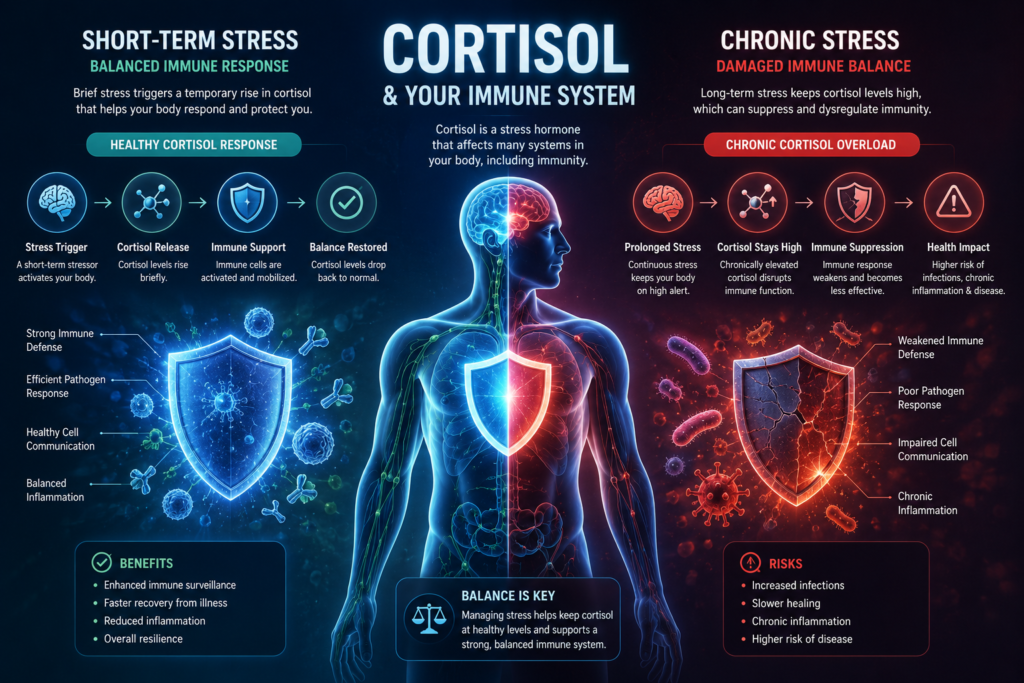
When discussing how stress affects immunity, it is critical to distinguish between acute stress and chronic stress. They do not affect the immune system in the same way; in fact, their effects are nearly opposite. The failure to understand this dichotomy often leads to confusion about whether stress “boosts” or “suppresses” immunity.
Acute Stress: The Evolutionary Advantage
Acute stress refers to short-term, highly intense stressors lasting minutes to hours. Examples include giving a public speech, taking a crucial exam, swerving to avoid a car accident, or, in ancestral terms, outrunning a predator.
During an acute stress event, the immune system actually experiences a temporary enhancement, specifically in natural (innate) immunity. From an evolutionary perspective, this makes perfect sense. If you are attacked by a predator, you are highly likely to suffer a wound, which means bacteria will breach your skin. To prepare for this, the sudden spike in adrenaline during an acute stressor causes a phenomenon called “lymphocyte redeployment.”
Billions of white blood cells (particularly Natural Killer cells and macrophages) detach from the lining of your blood vessels and are rapidly deployed to your skin, lymph nodes, and barrier tissues. The body is effectively moving its troops to the front lines in anticipation of injury and infection. Studies show that during acute stress, the body is highly primed to mount an aggressive, localized inflammatory response to heal wounds and fight off immediate bacterial threats. Once the threat passes, hormones drop, and the cells return to their normal resting state.
Chronic Stress: The Modern Trap
The problem arises when the stress response system is engaged continuously. Chronic stress refers to persistent, long-term challenges: an abusive relationship, systemic poverty, high-pressure corporate jobs, the prolonged illness of a family member, or persistent financial instability.
Because the human body did not evolve to sustain the “fight-or-flight” state indefinitely, chronic stress wreaks havoc on biological systems. The brain continuously perceives danger, keeping the HPA axis engaged and bathing the body in a steady stream of cortisol. As outlined by the Mayo Clinic, when the stress response stays turned on, overexposure to cortisol and other stress hormones disrupts almost all of the body’s processes, putting individuals at a significantly higher risk for chronic stress-related health problems.
Unlike acute stress, chronic stress is universally immunosuppressive. The continuous presence of cortisol tells the immune system to stand down, permanently reducing the number of circulating lymphocytes and halting the production of vital cytokines. The “troops” are not sent to the front lines; they are disbanded entirely.
Table 2: Acute vs. Chronic Stress and the Immune Response
| Feature | Acute Stress (Short-Term) | Chronic Stress (Long-Term) |
| Duration | Minutes to a few hours | Weeks, months, or years |
| Dominant Hormones | Adrenaline, Noradrenaline | Cortisol (Prolonged elevation) |
| Immune Effect | Immuno-enhancing (adaptive) | Immuno-suppressive (maladaptive) |
| Cellular Action | Redeployment of immune cells to skin/tissues | Depletion of circulating lymphocytes |
| Inflammatory Status | Localized, protective inflammation | Systemic, damaging chronic inflammation |
| Evolutionary Purpose | Survival against immediate physical injury | None; it is a modern biological mismatch |
Cellular and Molecular Mechanisms of Stress-Induced Immune Dysregulation
How exactly do stress hormones disarm the immune system at a microscopic level? Decades of rigorous inquiry have illuminated the complex cellular and molecular mechanisms driving stress-induced immune dysregulation.
1. Glucocorticoid Resistance and Systemic Inflammation
One of the most paradoxical and damaging effects of chronic stress involves the regulation of inflammation. Cortisol is naturally a potent anti-inflammatory hormone (which is why synthetic versions, like hydrocortisone and prednisone, are used to treat rashes and asthma). In a healthy body, cortisol binds to glucocorticoid receptors on immune cells, signaling them to stop producing inflammatory cytokines once an infection is cleared.
However, during chronic psychological stress, immune cells are constantly bombarded by high levels of cortisol. To protect themselves from this chemical deluge, the immune cells adapt by down-regulating their glucocorticoid receptors—a phenomenon known as glucocorticoid resistance. Because the cells become “deaf” to cortisol’s calming signals, they continue to produce pro-inflammatory cytokines unchecked. This results in a state of chronic, low-grade systemic inflammation, which is the root cause of countless modern diseases, including cardiovascular disease, type 2 diabetes, and metabolic syndrome.
2. The Th1 to Th2 Shift
Chronic stress fundamentally alters the balance of the adaptive immune system, specifically the ratio of Helper T-cells.
- Th1 Cells drive cellular immunity, orchestrating the defense against intracellular pathogens like viruses and certain bacteria.
- Th2 Cells drive humoral immunity, assisting B-cells in antibody production and managing defense against extracellular parasites (and are heavily involved in allergic responses).
Sustained neuroendocrine activation via cortisol and catecholamines suppresses the production of Th1 cytokines (like Interferon-gamma) while simultaneously upregulating Th2 cytokines (like Interleukin-4 and Interleukin-10). This “Th1-to-Th2 shift” means the stressed body becomes incredibly poor at fighting off viral infections (due to weakened Th1 responses) while simultaneously becoming hyper-reactive to allergens and prone to autoimmune attacks (due to overactive Th2 responses).
3. Beta-2 Adrenergic Receptor Signaling
As mentioned earlier, the sympathetic nervous system directly innervates lymphoid organs. The neurotransmitter noradrenaline binds directly to specific receptors on the surface of T-cells and B-cells, known as β2-adrenergic receptors. When chronic stress causes continuous engagement of these receptors, it severely impairs the ability of T-cells to multiply (proliferate) when they encounter a virus. Furthermore, it halts the maturation of dendritic cells, which are crucial antigen-presenting cells that show pathogens to T-cells to initiate an immune response.
4. Suppression of Natural Killer (NK) Cell Cytotoxicity
Natural Killer cells are vital for identifying and destroying cells that have become malignant (cancerous) or infected by viruses. Chronic psychological stress has been shown across numerous human and animal studies to dramatically reduce both the number of circulating NK cells and their functional cytotoxicity—meaning the remaining NK cells lose their ability to effectively kill targeted threats. This mechanism is one reason why chronic stress is heavily implicated in increased viral susceptibility and the progression of certain cancers.
Health Consequences of a Stressed Immune System
The molecular disruptions caused by chronic stress do not remain isolated in test tubes; they manifest as tangible, sometimes severe, health consequences in the human body. When the immune system is fundamentally handicapped by stress hormones, the biological fallout touches nearly every facet of human health. The Cleveland Clinic explicitly notes that “stress sickness” is a very real physiological state characterized by an overworked, exhausted immune system that can no longer adequately protect the host.
Increased Susceptibility to Infections
Perhaps the most thoroughly documented consequence of chronic stress is an increased vulnerability to viral and bacterial infections. In landmark studies conducted in the 1990s, researchers quarantined healthy volunteers, measured their baseline psychological stress levels, and then intentionally exposed them to the rhinovirus (the virus responsible for the common cold) via nasal drops. The results were undeniable: individuals who reported high levels of psychological stress in the months leading up to the study were significantly more likely to develop a clinical cold compared to their low-stress counterparts. The stress hormones had depleted their lymphocyte counts, leaving the mucosal barriers vulnerable to viral replication. This explains the common phenomenon of falling ill immediately after a period of intense work or a major life event.
Delayed Wound Healing
The immune system is the primary engine of tissue repair. Macrophages and inflammatory cytokines are required to clean out a wound, prevent infection, and stimulate the growth of new tissue. Studies examining individuals under chronic stress—such as caregivers for spouses with advanced dementia—have revealed startling findings. When researchers administered standardized, harmless puncture wounds to the skin of stressed caregivers and non-stressed control subjects, the wounds of the chronically stressed caregivers took up to 24% longer to heal completely. The sustained presence of cortisol suppressed the localized inflammatory response necessary for cellular regeneration.
Impaired Vaccine Efficacy
Vaccines work by introducing a harmless fragment of a pathogen to the adaptive immune system, prompting B-cells to create protective antibodies and memory cells. However, if the immune system is suppressed by stress at the time of vaccination, the body fails to mount a robust antibody response. Research has consistently shown that individuals experiencing chronic stress—such as loneliness, grief, or severe occupational strain—produce significantly fewer antibodies following influenza, hepatitis B, and even COVID-19 vaccinations. In some cases, the stress-induced immunosuppression is so severe that the vaccine fails to confer clinical protection entirely.
Autoimmune Diseases and Chronic Inflammation
While stress suppresses the body’s ability to fight external invaders, the paradoxical resulting systemic inflammation (due to glucocorticoid resistance) dramatically increases the risk of autoimmune conditions. Autoimmunity occurs when the immune system loses its tolerance and begins attacking the body’s own healthy tissues. Chronic unmanaged stress is heavily linked to the development, progression, and severe symptomatic flare-ups of numerous autoimmune diseases, including:
- Rheumatoid Arthritis: An autoimmune attack on the joints.
- Lupus (SLE): Systemic inflammation affecting skin, joints, kidneys, and the brain.
- Psoriasis: An immune-mediated acceleration of skin cell growth.
- Inflammatory Bowel Disease (IBD): Including Crohn’s disease and ulcerative colitis, driven by severe mucosal inflammation in the digestive tract.
In these conditions, periods of intense psychological distress almost invariably precede clinical flare-ups, highlighting the devastating impact of an imbalanced Th1/Th2 ratio and runaway inflammatory cytokines.
Actionable Strategies: Managing Stress to Boost Immunity

Understanding the grim physiological realities of chronic stress can, ironically, be quite stressful. However, the bidirectional nature of psychoneuroimmunology offers a profound silver lining: just as the mind can suppress the immune system, intentional psychological and behavioral interventions can reverse the damage, lower stress hormones, and rejuvenate immune function.
Protecting your immune system requires a proactive approach to down-regulating the HPA axis and shifting the autonomic nervous system out of the sympathetic “fight-or-flight” state and into the parasympathetic “rest-and-digest” state.
1. Mind-Body and Psychological Interventions
Because stress originates in the brain’s perception of a threat, changing that perception is the first line of defense.
- Cognitive Behavioral Therapy (CBT): Extensive research indicates that CBT—a therapeutic modality focused on identifying and altering negative thought patterns—can have measurable impacts on physical health. In studies involving immunocompromised individuals, such as those managing HIV, participation in CBT stress-management programs led to a documented reduction in viral load and an increase in crucial CD4+ T-cell counts, entirely independent of pharmaceutical changes.
- The Relaxation Response: Coined by Dr. Herbert Benson at Harvard University, the “relaxation response” is the physiological antithesis to the fight-or-flight response. It can be triggered through deep diaphragmatic breathing, progressive muscle relaxation, or meditation. Activating this response manually lowers heart rate, reduces blood pressure, and decisively halts the production of cortisol, giving the immune system the biochemical clearance it needs to function optimally.
- Mindfulness and Yoga: Practicing yoga acts as a dual intervention. The physical postures (asanas) combined with controlled breathing (pranayama) stimulate the vagus nerve, a major component of the parasympathetic nervous system. Furthermore, inverted poses are thought to assist in the physical circulation of lymphatic fluid, which houses white blood cells, aiding in the filtration of cellular waste and toxins.
2. Prioritizing Sleep Architecture
Sleep is not a luxury; it is a biological imperative and the most critical period for immune system maintenance. During the deepest stages of non-REM sleep, the HPA axis is at its quietest, and cortisol levels drop to their lowest point in the 24-hour circadian cycle. It is precisely during this low-cortisol window that the immune system performs its most vital work: consolidating “immunological memory” (helping T-cells and B-cells remember pathogens they encountered during the day) and releasing necessary cytokines for tissue repair. Chronic sleep deprivation artificially elevates daytime cortisol levels, mimicking a state of chronic psychological stress and dramatically lowering lymphocyte counts. Adults must prioritize 7 to 9 hours of high-quality sleep to maintain immunological integrity.
3. Nutritional Interventions
The gut microbiome—the trillions of bacteria residing in the human digestive tract—plays a central role in educating and regulating the immune system. Approximately 70% of the body’s immune cells reside in the gut-associated lymphoid tissue (GALT).
- Anti-inflammatory Diet: Diets high in refined sugars, trans fats, and ultra-processed foods provoke the immune system, adding fuel to the fire of stress-induced systemic inflammation. Conversely, a diet rich in whole grains, lean proteins, omega-3 fatty acids (found in oily fish), and diverse plant fibers provides the necessary raw materials for immune cells to function.
- Microbiome Support: Fiber acts as a prebiotic, feeding beneficial gut bacteria which, in turn, produce short-chain fatty acids (SCFAs). SCFAs cross the gut barrier and act as powerful systemic anti-inflammatories, helping to counteract the damage caused by stress hormones.
4. Strategic Physical Exercise
Moderate, consistent physical exercise is a potent immune modulator. Cardiovascular exercise increases blood circulation, allowing immune cells to patrol the body more efficiently. It also briefly elevates body temperature, which can help inhibit bacterial growth, and effectively “flushes” the lungs and airways of potential pathogens. Most importantly, regular exercise lowers baseline levels of stress hormones over time and stimulates the release of endorphins—the brain’s natural mood elevators and painkillers. However, balance is crucial; extreme, unremitting physical exertion (like running back-to-back ultramarathons without adequate recovery) can mimic chronic stress, causing a temporary, severe suppression of the immune system.
5. Fostering Social Support
Human beings are inherently social creatures. Evolutionary biology suggests that isolation was historically a massive survival threat, which is why the modern experience of loneliness is registered by the brain as a chronic stressor, elevating cortisol and inflammation. Conversely, strong social bonds, positive relationships, and community engagement release oxytocin. Oxytocin, often dubbed the “love hormone,” directly antagonizes cortisol, lowering blood pressure and dampening the reactivity of the HPA axis. Cultivating a strong support network is not just emotionally fulfilling; it is a biological shield that protects your immune cells from the ravages of psychological stress.
Conclusion
The intersection of the mind and the immune system represents one of the most fascinating frontiers in modern medicine. The science of psychoneuroimmunology has definitively proven that the brain and the immune system are not isolated silos, but rather intimately connected partners engaged in a continuous, lifelong dialogue.
Stress is an unavoidable facet of the human experience, and our bodies are beautifully equipped to handle it in short, acute bursts. The danger arises when the pressures of modern life trap us in a perpetual state of biological alarm. Chronic stress acts as a corrosive force, flooding the body with cortisol and catecholamines, driving dangerous systemic inflammation, silencing critical immune cells, and leaving the body vulnerable to infections, delayed healing, and autoimmune disease.
However, understanding this physiology is profoundly empowering. Because the communication pathway is bidirectional, we hold the power to influence our internal biology. By acknowledging the heavy toll that psychological strain places on our physical defenses, we can elevate stress management from a secondary luxury to a primary health necessity. Through intentional practices—whether it is cognitive behavioral therapy, deep breathing, prioritizing restorative sleep, engaging in moderate exercise, or cultivating deep social connections—we can actively send signals of safety to our brains. In doing so, we quiet the alarm bells of the HPA axis, lower the tide of cortisol, and allow our immune system to return to its natural, vigilant state, ready to protect us as it was evolved to do.
Emily Carter is a Senior Health Researcher and Supplement Analyst at the Nutrasfit Research Team, based in Austin, Texas. She specializes in evaluating dietary supplements through ingredient analysis, scientific research, and real-world effectiveness.
With a background in nutrition science, Emily focuses on breaking down complex health information into simple, practical insights that readers can trust. Her work is centered on helping individuals make informed decisions and choose supplements that are safe, effective, and aligned with their health goals.