
In recent years, the scientific and medical communities have undergone a profound paradigm shift in their understanding of human health. At the very center of this revolution is the gut microbiome—a complex, bustling, and invisible ecosystem residing primarily in our large intestine. Comprising trillions of microorganisms, this dynamic environment is not merely a passive recipient of the food we consume; it is an active, central command center that dictates our physical, mental, and metabolic well-being. From regulating the immune system to synthesizing essential neurotransmitters like serotonin and dopamine, the state of your gut is inextricably linked to your overall vitality.
However, modern dietary habits have introduced unprecedented challenges to this delicate ecosystem. The standard western diet, characterized by convenience, high caloric density, and heavy industrial processing, has fundamentally altered the microbial landscape of the human digestive tract. While a significant amount of attention is rightly given to the foods we should eat to support gut health—such as probiotic-rich fermented foods and prebiotic fibers—it is equally, if not more, critical to understand the foods that actively damage it.
This comprehensive guide delves deep into the physiological mechanisms of gut health and systematically breaks down the common, everyday foods and additives that disrupt the microbiome, degrade the intestinal barrier, and promote systemic inflammation.
Part 1: The Physiology of a Healthy Gut Ecosystem
To truly understand how certain foods damage the gut, one must first grasp the intricate biological architecture of a healthy intestinal environment. The human gut is often referred to as the “second brain” and the body’s primary defense system, and this is entirely due to the synergistic relationship between our human cells and our resident microbial populations. For an introductory look at the immense scale of this ecosystem, you can explore Wikipedia’s overview of the gut microbiome.
The Intestinal Barrier: The Ultimate Gatekeeper
The inner lining of the gastrointestinal tract is remarkably thin—in most places, it is only a single layer of epithelial cells thick. This microscopic wall has a dual, seemingly contradictory mandate: it must be permeable enough to allow vital nutrients, vitamins, and water to pass into the bloodstream, yet secure enough to keep out pathogens, toxins, and undigested food particles.
This balancing act is achieved through structures known as tight junctions. Made of specialized proteins like claudins and occludins, tight junctions act as the cellular “glue” that binds adjacent epithelial cells together, sealing the spaces between them. When these tight junctions function correctly, the gut is secure. When they are compromised, the barrier becomes hyper-permeable, leading to a condition colloquially known as “leaky gut” or, scientifically, intestinal permeability. For further clinical context, refer to Cleveland Clinic’s guide to leaky gut syndrome.
The Mucosal Layer: The First Line of Defense
Above the epithelial cells lies a thick, viscous layer of mucus. This mucus is divided into two distinct strata:
- The Inner Layer: This layer is densely packed and practically sterile. It acts as a physical buffer, preventing bacteria from coming into direct contact with the vulnerable epithelial cells.
- The Outer Layer: This layer is looser and serves as a nutrient-rich habitat for specific commensal (friendly) bacteria, such as Akkermansia muciniphila. These bacteria feed on the mucins (proteins that make up mucus) and, in return, stimulate the host body to produce more mucus, ensuring the barrier remains robust.
Short-Chain Fatty Acids (SCFAs): The Currency of Gut Health
When beneficial bacteria in the colon ferment dietary fibers—specifically complex carbohydrates that human digestive enzymes cannot break down—they produce metabolic byproducts known as Short-Chain Fatty Acids (SCFAs). The most important of these are acetate, propionate, and butyrate.
Butyrate is the primary energy source for colonocytes (the cells lining the colon). It possesses profound anti-inflammatory properties, helps maintain the integrity of tight junctions, and keeps the pH of the colon slightly acidic, which creates an inhospitable environment for pathogenic bacteria. The Harvard T.H. Chan School of Public Health offers an excellent overview on how diet influences these metabolic processes in their article on the Microbiome.
Dysbiosis: When the Ecosystem Collapses
When the balance between beneficial, SCFA-producing bacteria and harmful, pro-inflammatory microbes tips in favor of the latter, a state of dysbiosis occurs. Dysbiosis is characterized by a loss of microbial diversity, the overgrowth of opportunistic pathogens, and the degradation of the mucosal layer. This state is the precursor to chronic, low-grade systemic inflammation, which is the biological root of numerous modern afflictions, including metabolic syndrome, autoimmune diseases, and neurodegenerative conditions.
Part 2: Ultra-Processed Foods (UPFs) and Dietary Emulsifiers
Ultra-processed foods (UPFs) now account for the majority of caloric intake in many Western nations. These foods—which include packaged snacks, commercially baked goods, reconstituted meats, and sugary cereals—are engineered for hyper-palatability, long shelf-life, and cost-efficiency. However, the very ingredients that make UPFs profitable make them profoundly destructive to the gut microbiome.
The Hidden Danger of Emulsifiers
One of the most insidious categories of food additives found in UPFs are dietary emulsifiers. In culinary chemistry, oil and water do not mix. Emulsifiers are molecules with both a hydrophilic (water-loving) and hydrophobic (oil-loving) end, allowing them to bind fat and water together into a stable, smooth emulsion. This is why commercial mayonnaise doesn’t separate in the jar and why mass-produced ice cream feels exceptionally smooth on the palate.
While regulatory bodies historically classified these additives as “Generally Recognized As Safe” (GRAS) based on acute toxicity tests, modern microbiome research has revealed a much darker picture. Extensive studies have shown that common dietary emulsifiers—specifically Carboxymethylcellulose (CMC), Polysorbate 80 (P80), and Carrageenan—wreak havoc on the intestinal environment.
The Detergent Effect
Because emulsifiers are essentially chemical detergents, they interact with the gut environment in much the same way dish soap interacts with grease. When ingested, these compounds scour the gastrointestinal tract, disrupting the delicate mucosal layer that protects the epithelial lining.
Research demonstrates that exposure to CMC and P80 leads to a severe thinning of the mucus gut barrier. Without this physical barrier, gut bacteria are brought into direct, inappropriate contact with the epithelial cells. This triggers a localized immune response, resulting in gut inflammation.
Microbial Shifts and Pathogen Promotion
Emulsifiers do not just damage human tissue; they directly alter the composition of the microbiome. In vitro and in vivo studies have confirmed that exposure to synthetic emulsifiers leads to a sharp decline in beneficial bacterial populations, such as Faecalibacterium prausnitzii (a key butyrate producer) and Akkermansia muciniphila (the crucial mucus-regulating bacterium).
Simultaneously, these additives selectively promote the growth of pro-inflammatory, potentially pathogenic bacteria. The shift in microbial architecture results in a massive reduction in the production of protective SCFAs, leaving the colon vulnerable. Over prolonged periods, this emulsifier-induced dysbiosis has been closely linked to the development of metabolic syndrome, obesity, Irritable Bowel Syndrome (IBS), and Functional Dyspepsia (FD).
Table 1: Common Dietary Additives and Their Impact on Gut Health
| Additive Name | Common Food Sources | Identified Impact on the Gut Microbiome & Intestinal Barrier |
| Carboxymethylcellulose (CMC / E466) | Ice cream, baked goods, diet foods, sauces | Acts as a detergent; thins the protective mucus layer; promotes low-grade gut inflammation; alters bacterial motility. |
| Polysorbate 80 (P80 / E433) | Margarine, chewing gum, commercial condiments | Increases abundance of pathogenic Enterobacteriaceae; decreases beneficial Bacteroidaceae; linked to weight gain and metabolic syndrome in animal models. |
| Carrageenan (E407) | Almond milk, deli meats, yogurt, infant formula | Triggers gut inflammation; reduces levels of Bifidobacterium; increases production of pro-inflammatory cytokines; disrupts gut barrier. |
| Titanium Dioxide (E171) | Candies, chewing gum, white-colored sauces | Nano-particles can accumulate in the gut lining; impairs intestinal barrier function; increases inflammation and reduces bacterial diversity. |
| High Fructose Corn Syrup (HFCS) | Sodas, sweet snacks, fast food | Overwhelms small intestine absorption, leading to colonic fermentation; promotes the overgrowth of opportunistic fungi and harmful bacterial strains. |
Part 3: Artificial Sweeteners: The Zero-Calorie Illusion
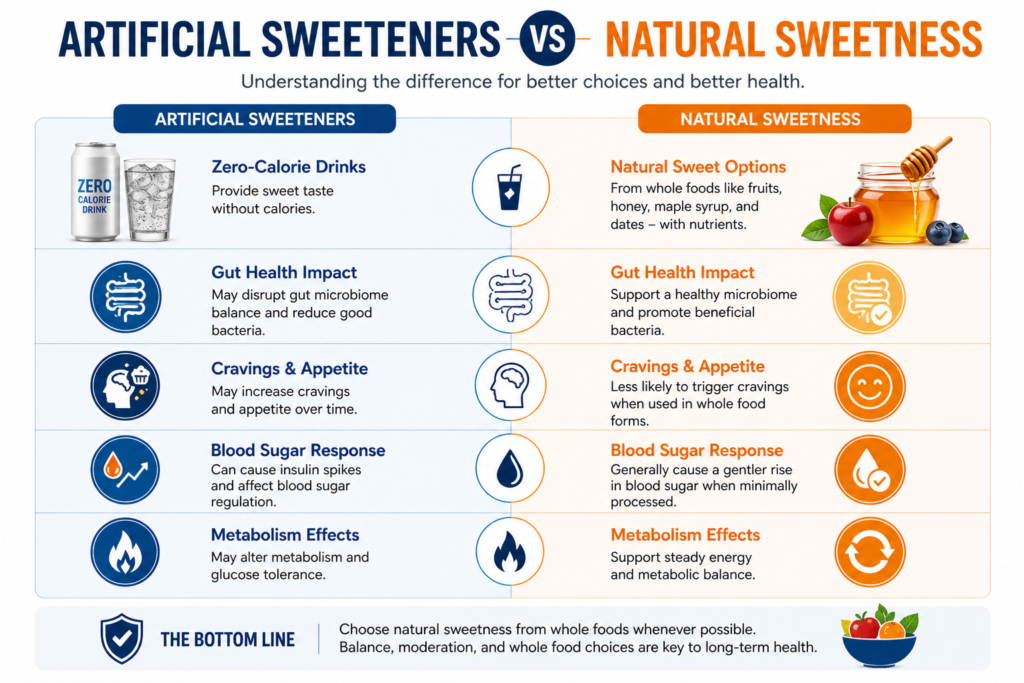
In a bid to reduce caloric intake and combat obesity, millions of people have turned to non-caloric artificial sweeteners (NAS) and sugar substitutes. Marketed as guilt-free alternatives, chemicals like Sucralose, Aspartame, Saccharin, and Acesulfame Potassium (Ace-K) are ubiquitous in diet sodas, protein powders, sugar-free gum, and baked goods.
For decades, the prevailing nutritional dogma was that because these compounds pass through the human digestive system without being metabolized for energy, they are biologically inert. Recent scientific literature has decisively debunked this myth. While they may not provide calories to human cells, they actively interact with our microbial residents.
Microbial Disruption and Pro-Inflammatory Genetics
Because many artificial sweeteners are not absorbed in the upper gastrointestinal tract, they arrive in the colon fully intact. Here, they encounter the microbiome.
Research into sucralose (one of the most widely consumed artificial sweeteners globally) has shown that chronic consumption severely alters the developmental dynamics of the gut microbiome. While short-term use might not show immediate catastrophe, long-term exposure fundamentally shifts the bacterial composition. More alarmingly, studies utilizing 16S rRNA gene sequencing have revealed that sucralose consumption significantly increases the abundance of bacterial genes related to pro-inflammatory mediators.
Specifically, artificial sweeteners have been shown to upregulate the bacterial synthesis of Lipopolysaccharides (LPS). LPS are large molecules found on the outer membrane of Gram-negative bacteria; they are highly toxic endotoxins. When LPS production increases and the gut barrier is compromised, these toxins flood into the bloodstream, triggering systemic, chronic inflammation. The presence of artificial sweeteners also increases the expression of bacterial flagella and fimbriae synthesis—structures that allow bacteria to move and adhere to the intestinal wall, often a sign of increased pathogenicity.
The Glucose Intolerance Paradox
Perhaps the most ironic and devastating consequence of artificial sweetener consumption is its effect on metabolic health. While people consume NAS to manage blood sugar and prevent diabetes, the microbial dysbiosis caused by these chemicals actually induces glucose intolerance.
Landmark microbiome research has demonstrated that when artificial sweeteners alter the gut bacteria, those altered microbial communities begin sending faulty signals to the host’s metabolism. In controlled trials, individuals consuming high amounts of saccharin and sucralose developed poor blood sugar responses. When the fecal microbiota from these individuals was transplanted into germ-free mice, the mice rapidly developed identical glucose intolerance and insulin resistance. The artificial sweetener itself did not spike the blood sugar; rather, the damaged microbiome triggered metabolic dysfunction. To understand how these sweeteners are generally viewed in clinical settings, you can review Mayo Clinic’s advice on artificial sweeteners.
Part 4: Red Meat, Processed Meats, and the TMAO Pathway
The debate over red meat consumption has raged for decades, often centering around saturated fat and cholesterol. However, the lens of the microbiome has provided a completely new, metabolically complex perspective on how heavy red meat consumption damages cardiovascular and intestinal health.
Red meats (such as beef, lamb, and pork) and processed meats (like bacon, sausages, and hot dogs) are highly abundant in specific nutrients, primarily L-carnitine and Choline. While these nutrients are essential in small amounts, their overabundance in a meat-heavy diet initiates a dangerous biochemical cascade orchestrated by gut bacteria.
The TMAO Production Pathway
When you consume a steak or a heavy serving of bacon, the L-carnitine and choline are delivered to the colon. Certain strains of gut bacteria utilize these compounds as fuel sources. As the bacteria metabolize the L-carnitine and choline, they release an intermediate gas called Trimethylamine (TMA).
The TMA gas is rapidly absorbed through the intestinal wall into the portal circulation, where it is carried directly to the liver. In the liver, an enzyme known as flavin monooxygenase 3 (FMO3) oxidizes the TMA, converting it into a molecule called Trimethylamine-N-oxide (TMAO). For an in-depth chemical breakdown of this molecule, see Wikipedia’s entry on Trimethylamine N-oxide (TMAO).
The Cardiovascular and Endothelial Consequences
TMAO is highly toxic to the vascular system. Elevated circulating levels of TMAO are strongly correlated with an increased risk of severe cardiovascular disease, heart attacks, and strokes. TMAO promotes the formation of macrophage foam cells—the primary cells responsible for the buildup of atherosclerotic plaque in the arteries.
Furthermore, TMAO induces vascular inflammation, promotes oxidative stress, and causes endothelial dysfunction (stiffening and malfunctioning of the blood vessels). It even increases platelet hyperreactivity, making the blood more prone to clotting.
What makes this mechanism uniquely fascinating is its absolute reliance on the microbiome. Studies have shown that strict vegans and vegetarians, who possess a different microbial profile due to their plant-based diets, do not produce significant levels of TMAO even if they are temporarily fed L-carnitine supplements. Their microbiomes simply lack the specific bacterial strains required to convert the meat nutrients into toxic TMA. Conversely, long-term, high-volume consumption of red meat actively selects for and breeds the very bacteria that produce TMA, creating a vicious cycle of toxicity.
In addition to the L-carnitine issue, processed meats contain high levels of artificial preservatives, namely nitrates and nitrites. While these keep meat looking red and fresh, they undergo chemical reactions in the gut that form nitrosamines, which are well-documented carcinogens linked heavily to the development of colorectal cancer, further compounding the gastrointestinal damage.
Part 5: Alcohol Consumption and the Enterohepatic Axis
While occasional, moderate alcohol consumption (particularly polyphenol-rich red wine) is often cited as benign or even mildly beneficial, regular or excessive alcohol intake is an aggressive antagonist to the gastrointestinal tract. Alcohol (ethanol) exerts a dual wave of destruction: it causes direct physical toxicity to the tissues and orchestrates a massive disruption of the microbial balance.
Direct Toxicity and the Breakdown of the Barrier
When alcohol enters the gastrointestinal tract, a significant portion is absorbed in the small intestine. Ethanol is a potent solvent; it directly damages the mucosal epithelium in a dose-dependent manner. Alcohol consumption dismantles the microtubule skeletons of intestinal cells, induces severe oxidative stress, and directly destabilizes the F-actin cytoskeleton.
Crucially, alcohol forcefully disrupts the tight junction proteins (the “glue” holding the intestinal barrier together). This leads to rapid and profound intestinal hyperpermeability (leaky gut). Acute binge drinking, even in healthy adults, has been shown to cause an immediate spike in blood serum endotoxin levels, proving that the gut barrier has been successfully breached.
Microbiome Shifts and the Gut-Liver Axis
Beyond physical damage, alcohol dramatically alters the microbiome. Chronic alcohol consumption suppresses Paneth cells, specialized cells in the intestinal lining that secrete antimicrobial peptides. With the intestine’s innate defense system down-regulated, opportunistic and pathogenic bacteria begin to overgrow and thrive.
Alcohol triggers a steep decline in the abundance of protective, SCFA-producing families like Bifidobacteria and Lactobacilli. This lack of butyrate starves the colon cells of their primary energy source, exacerbating the breakdown of the gut lining.
Because the barrier is compromised, the massive loads of toxic Lipopolysaccharides (LPS) produced by the overgrown pathogenic bacteria leak into the bloodstream. Because of the body’s anatomy, blood from the intestines drains directly into the liver via the portal vein. This flood of endotoxins hits the liver, activating hepatic stellate cells and triggering intense liver inflammation. This continuous bombardment of the liver by gut-derived toxins is the primary mechanism behind Alcoholic Liver Disease (ALD), liver fibrosis, and eventual cirrhosis. The gut and the liver operate on an axis, and when alcohol destroys the gut ecosystem, the liver pays the ultimate physiological price.
Part 6: Refined Carbohydrates, Added Sugars, and the High-Fat Dilemma
The final pillar of gut-damaging foods revolves around macro nutrient imbalances, specifically the combination of refined carbohydrates, massive doses of added sugar, and low-quality fats, paired with a devastating lack of dietary fiber.
Starving the Good Bacteria
A healthy microbiome requires complex, fermentable carbohydrates—often referred to as Microbiota-Accessible Carbohydrates (MACs)—to survive. These are found in vegetables, legumes, whole grains, and fruits. Refined grains (like white flour, white rice, and commercial pastries) have had their fibrous structures completely stripped away during milling.
When you consume a diet high in refined carbohydrates and low in dietary fiber, you are effectively starving your beneficial bacteria. Driven by starvation, certain bacterial species are forced to change their feeding habits. In the absence of dietary fiber, mucin-degrading bacteria will turn to the host’s own mucus lining as a food source. They literally begin to eat the protective mucosal layer away, moving dangerously close to the epithelial wall and triggering an inflammatory immune response.
The Sugar and Pathogen Feed
Added sugars—particularly high fructose corn syrup and refined white sugar—pose a different problem. While complex carbohydrates take time to break down, feeding bacteria in the lower colon, refined sugars are rapidly absorbed. However, when consumed in the massive quantities typical of modern diets, the small intestine’s absorptive capacity is overwhelmed.
The excess sugar spills into the large intestine, where it acts as explosive rocket fuel for opportunistic pathogens and fungi. High-sugar diets are heavily linked to the overgrowth of Candida albicans (a yeast species) and pro-inflammatory bacterial strains. This sudden, rapid fermentation creates excessive gas, bloating, and osmotic diarrhea.
Unhealthy Fats and Bile Acid Toxicity
Dietary fats interact with the microbiome via the biliary system. When you consume high amounts of poor-quality saturated fats (from deep-fried foods, fast food, and industrialized seed oils high in Omega-6s), the liver is forced to secrete massive amounts of primary bile acids to emulsify the fat.
While bile acids are necessary for fat digestion, an overabundance of them entering the colon is highly disruptive. Certain pathogenic gut bacteria possess enzymes that convert these primary bile acids into secondary bile acids (such as deoxycholic acid and lithocholic acid). Secondary bile acids are highly toxic to human cells; they cause oxidative DNA damage, promote cellular proliferation, and are heavily implicated in the promotion of colorectal cancer. Furthermore, diets excessively high in Omega-6 polyunsaturated fats (commonly found in cheap frying oils) directly promote systemic inflammation, contrasting sharply with the anti-inflammatory properties of Omega-3 fats found in fish and flaxseeds.
Part 7: Identifying Gut Damage – Signs, Symptoms, and Diagnostics
Because the gut microbiome is involved in virtually every physiological system, the symptoms of a damaged gut extend far beyond simple digestion. Recognizing the signs of dysbiosis and intestinal permeability is crucial for early intervention.
Gastrointestinal Symptoms:
- Chronic bloating, excessive flatulence, and abdominal distension.
- Erratic bowel movements, oscillating between diarrhea and constipation.
- Diagnosis of Irritable Bowel Syndrome (IBS), Inflammatory Bowel Disease (IBD), or Functional Dyspepsia.
- Acid reflux and persistent heartburn.
Systemic and Neurological Symptoms:
- Brain Fog and Fatigue: Due to neuroinflammation caused by circulating endotoxins (LPS) that have breached the blood-brain barrier.
- Skin Conditions: The “gut-skin axis” means dysbiosis frequently presents as acne, eczema, rosacea, and persistent dermatitis.
- Autoimmune Conditions: Leaky gut allows undigested food particles into the bloodstream, where the immune system attacks them, potentially triggering molecular mimicry and autoimmune flare-ups (e.g., Rheumatoid arthritis, Hashimoto’s thyroiditis).
- Mood Disorders: With the majority of the body’s serotonin produced in the gut, dysbiosis is heavily linked to sudden onsets of anxiety, depression, and mood volatility.
Table 2: The Microscopic Profile of a Healthy vs. Damaged Gut
| Physiological Metric | The Healthy, Thriving Microbiome | The Damaged, Dysbiotic Microbiome |
| Microbial Diversity | High diversity; rich variety of species coexisting in balance. | Low diversity; dominance of a few opportunistic, pathogenic strains. |
| SCFA Production | High levels of butyrate, acetate, and propionate. | Drastically reduced SCFA synthesis; colonocytes starved of energy. |
| Mucosal Layer | Thick, dense, and continually regenerated; physically separates bacteria from cells. | Thinned, degraded, or entirely stripped away by emulsifiers or starving bacteria. |
| Tight Junctions | Securely closed; highly selective permeability. | Broken or lax (leaky gut); allowing toxins and large molecules into the bloodstream. |
| Systemic Immune State | Calm, regulated, and tolerant. | Hyperactive, inflamed, continuously reacting to endotoxin (LPS) leakage. |
| Dominant Bacteria Types | Bifidobacterium, Lactobacillus, Faecalibacterium, Akkermansia. | Enterobacteriaceae, Desulfovibrio, elevated levels of Staphylococcus and Streptococcus. |
Part 8: Actionable Strategies for Microbiome Restoration
Understanding what damages the gut is only half the battle. If your microbiome has been compromised by years of ultra-processed foods, artificial sweeteners, excessive red meat, or alcohol, the human body is remarkably resilient. The microbiome is highly plastic, meaning it can be reshaped and restored through deliberate, consistent dietary interventions.
1. The Elimination Phase
You cannot heal a wound while continuously picking at it. The first step in gut restoration is significantly reducing or entirely eliminating the primary offenders:
- Audit your pantry: Read ingredient labels obsessively. Throw out anything containing Carboxymethylcellulose (CMC), Polysorbate 80, Carrageenan, and artificial sweeteners (Sucralose, Aspartame, Saccharin).
- Moderate Meat and Alcohol: Reduce red meat consumption to 1-2 times per week, opting for lean cuts. Substitute with high-quality poultry, fish, or plant-based proteins to lower TMAO production. Limit alcohol intake strictly.
- Cut the Sugar: Eliminate refined, added sugars to starve out pathogenic yeast and opportunistic bacteria.
2. Introduce Prebiotics (Fertilizer for the Gut)
Prebiotics are the non-digestible fibers that specifically feed beneficial bacteria. Without prebiotics, probiotics cannot thrive. You must introduce high quantities of Microbiota-Accessible Carbohydrates (MACs).
- Inulin and Fructooligosaccharides (FOS): Found abundantly in garlic, onions, leeks, asparagus, and Jerusalem artichokes.
- Resistant Starch: A highly effective prebiotic that resists digestion in the small intestine. Found in green (unripe) bananas, cooked and cooled potatoes, and cooked and cooled rice.
3. Embrace Polyphenols (Microbial Modulators)
Polyphenols are powerful plant-based antioxidants that humans struggle to digest, but which gut bacteria love. Polyphenols act as regulators, inhibiting the growth of pathogenic bacteria while stimulating the growth of beneficial strains like Bifidobacteria.
- Sources: Wild blueberries, blackberries, dark chocolate (85%+ cacao), green tea, cloves, and extra-virgin olive oil.
4. Probiotics and Fermented Foods (The Reinforcements)
While taking a probiotic pill can be helpful, acquiring transient beneficial microbes through fermented foods is a more biologically natural approach. These foods introduce millions of live, active cultures that help acidify the gut and crowd out pathogens.
- Sources: Unpasteurized sauerkraut, traditional kimchi, milk kefir, water kefir, and unsweetened kombucha. Note: Ensure these are naturally fermented and kept in the refrigerated section of the grocery store, as shelf-stable equivalents have had their bacteria killed off by heat.
Conclusion
The human gut microbiome is an exquisite, highly sensitive ecosystem that requires constant maintenance and respect. For the vast majority of human history, our diets consisted of whole, unprocessed, fiber-rich foods that naturally cultivated a robust and diverse bacterial landscape. The advent of modern food processing, artificial additives, synthetic sweeteners, and excessive alcohol consumption represents an unprecedented assault on our internal biology.
Foods that damage gut health do not merely cause temporary digestive discomfort; they fundamentally alter our microbial genetics, strip away our internal physical barriers, and open the floodgates to chronic, systemic inflammation. The connection between the breakdown of the gut wall and the rise of metabolic syndrome, cardiovascular disease, autoimmune conditions, and mental health disorders is no longer a fringe theory—it is a scientifically established reality.
By becoming a conscious consumer, reading labels to avoid emulsifiers, rejecting the false promise of artificial sweeteners, moderating red meat and alcohol, and prioritizing whole, fiber-rich foods, you can reclaim control over your microbiome. Healing the gut is a gradual, deliberate process, but it is unequivocally the most powerful intervention you can make for your long-term health and vitality.
Further Reading & Scientific Sources
The scientific mechanisms, microbial analyses, and metabolic pathways detailed in this guide are synthesized from contemporary medical and microbiological research. For further exploration of the peer-reviewed literature regarding food additives, the TMAO pathway, artificial sweeteners, and the gut-liver axis, please consult the primary literature databases below:
- On Emulsifiers: Research published in Frontiers in Microbiology (2020) outlines how synthetic emulsifiers like CMC and Polysorbate 80 severely impact gut microbiota directly, lowering microbial diversity and altering short-chain fatty acid production irrespective of their origins.
- On Artificial Sweeteners: Studies available via Frontiers in Physiology and MDPI highlight the mechanisms by which sucralose and saccharin alter the developmental dynamics of the microbiome, increase the synthesis of bacterial pro-inflammatory genes (like LPS), and subsequently trigger glucose intolerance.
- On Red Meat & TMAO: Extensive literature available through the National Institutes of Health (NIH) and PMC details how intestinal microbiota metabolize dietary L-carnitine and choline from red meat to produce Trimethylamine-N-oxide (TMAO), a potent driver of vascular inflammation and accelerated atherosclerosis.
- On Alcohol & Leaky Gut: Comprehensive reviews in PMC and NIH databases explain the specific mechanisms by which ethanol abuse causes acute damage to the colonic epithelial barrier, suppresses Paneth cell immunity, and drives hepatic fibrosis through the gut-liver axis.
Emily Carter is a Senior Health Researcher and Supplement Analyst at the Nutrasfit Research Team, based in Austin, Texas. She specializes in evaluating dietary supplements through ingredient analysis, scientific research, and real-world effectiveness.
With a background in nutrition science, Emily focuses on breaking down complex health information into simple, practical insights that readers can trust. Her work is centered on helping individuals make informed decisions and choose supplements that are safe, effective, and aligned with their health goals.